Research:
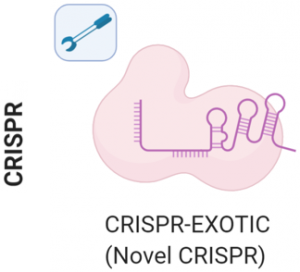
1. Discovery and engineering of novel CRISPR/Cas systems
We are interested in discovering and engineering novel CRISPR/Cas systems towards improving the repertoire for gene editing, gene correction, and diagnostics. Our lab is also investigating the mechanism of various Type II, V, and VI CRISPR systems.
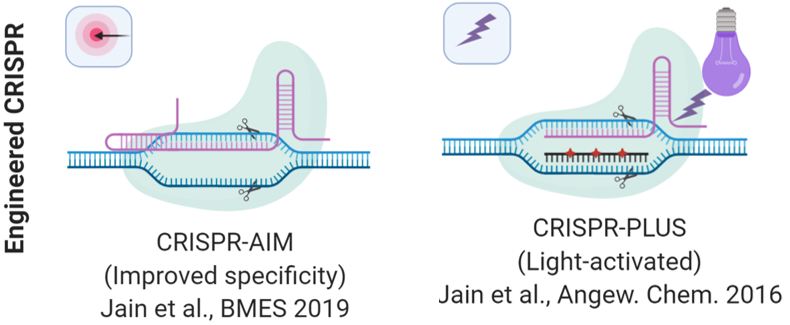
2. Enhanced sensitivity, specificity, and control of CRISPR/Cas systems
We focus on understanding the CRISPR/Cas mechanisms to engineer novel genome editing tools with improved activity, specificity, sensitivity, and function. To edit genes in precise location and time, we created a light-activatable CRISPR-PLUS (CRISPR-precise light-mediated unveiling of sgRNAs) system by incorporating a photocleavable complementary oligonucleotide to the guide RNA, which allowed precise control of genome editing with light. We also demonstrated precise control of CRISPR activity with multiple different sgRNAs concurrently using CRISPR-PLUS in vitro enabling the multiplexing capabilities of CRISPR-PLUS (Jain et al., Angew. Chem. 2016). We recently discovered that CRISPR-PLUS demonstrates significant improvement in specificity of CRISPR, which is currently being investigated in our lab.
In addition, we also discovered various modifications on crRNAs that can drastically enhances the sensitivity and specificity of CRISPR/Cas systems (Nguyen et al., Nature Communications, 2020).
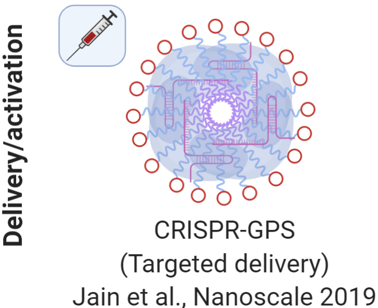
3. Targeted delivery of CRISPR/Cas systems with improved genomic integration
We are engineering both non-viral and viral vectors for delivery for efficient editing and genome integration.
Recently we developed an efficient targeted delivery system in vitro and in vivo, using guiding peptide sequences (GPS) carrying a lipid tail, cell-penetrating domain, and a targeting domain. When mixed with CRISPR/Cas, we discovered that these peptide-lipids form nanocomplexes electrostatically (CRISPR-GPS) and can deliver the complexes to a variety of cell lines and can be potentially applied in vivo (Jain et al., Nanoscale 2019).
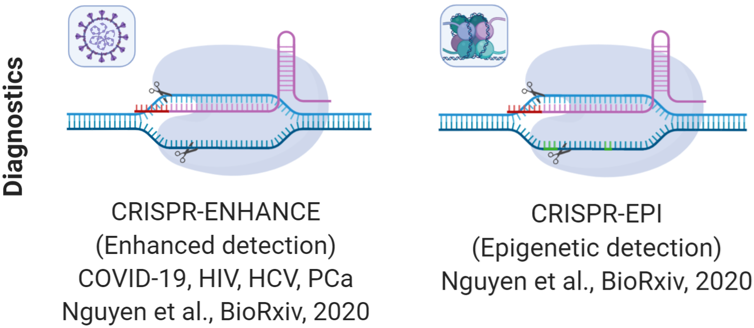
4. CRISPR/Cas as improved diagnostics
We recently discovered that modified crRNAs for LbCas12a systems that can increase the speed of trans-cleavage by 3.2-fold, making it one of the fastest reported CRISPR/Cas in terms of trans-cleavage activity and we termed it CRISPR-ENHANCE. Using CRISPR-ENHANCE, we observed up to femtomolar sensitivity of nucleic acid detection without any target amplification. We applied the technology for improved detection of #SARSCoV-2 RNA using paper-based diagnostics as well as for detecting prostate cancer, HIV & HCV genes. We also discovered a significant improvement in specificity of detection by several-fold (up to 8.8-fold) with CRISPR-ENHANCE(Nguyen et al., Nature Communications, 2020).
Funding: Check out grants
Facilities:
Our lab’s expertise in chemically modifying and testing nucleic acids, peptides, and proteins intersects chemistry, biology, nanotechnology and genomics.
- Chemistry:Chemical modification, purification and characterization of peptides, oligonucleotides, polymers, and small molecules. Jain lab has a dedicated chemistry suite with a hood and equipment.
- We own a state-of-the-art microwave-assisted peptide synthesizer (CEM’s Liberty Blue), a HPLC (Agilent 1100) for purification, a NanoDrop for UV/Vis (Thermo oneC), a fluorimeter with 384-well capabilities (Biotek), UV crosslinker, a large gravity oven, and various small equipment. Easy access to LC/MS/MS analysis in the proteomics core within the building.
- Protein Engineering: Jain Lab has an entire pipeline for discovering and engineering proteins. Well equipped with bioinformatics pipeline, plasmid design and reconstruction, recombinant DNA, protein expression and purification.
- We have Two FPLCs (GE Akta Pure and BioRad Biologic) with affinity, SEC, and IEC set ups, multiple temperature-controlled shakers and centrifuges, incubators, autosonifier, and homogenizer within the lab. Several shared ultracentrifuges, cold room, and dark room on the same floor. Easy access to LC/MS, MALDI, monoclonal antibody production, and bio-layer interferometry as a part of the core facilities within the building. Access to NMR, circular dichroism, and X ray crystallography on campus.
- Nanotechnology: Jain lab is set up for developing and analyzing a variety of nanoparticles and liposomal formulations.
- DLS/Zetasizer (coming soon). Easy access to NanoSight NS300 and various electron microscopes within the building. A range of nano-characterization equipment available at the Research Service Center on campus.
- Liquid Handling: Fast and safe liquid transfer
- We have an automated liquid handler (Opentron OT-2) with single and multichannel options, Electronic multichannel pipettes (Integra and Thermo) and various other pipettors for liquid dispensing.
- Molecular Biology: Jain lab is set up for a variety of molecular biology techniques including qPCR, PCR, plasmid design, siRNA design, sgRNA design and screening, in vitro transcription, gel electrophoresis, Western blot, ELISA, and enzymatic assays
- Two qPCR (Agilent and ViiA 7 systems) and three PCR machines (Thermo 3-block, Eppendorf, and BioRad), an iWestern workflow system (Thermo), and several gel electrophoresis systems. We also own two gel imagers- a phosphorimager/chemiluminescent/fluorescence (GE Typhoon) and a RGB gel imager (Analytik Jena, GelStudio Touch Plus). A variety of liquid handling and small molecular biology equipment are also in the lab.
- Tissue Culture: Jain lab is equipped with mammalian cell culture, transfection, transduction, transformation, electroporation, viral production, and microscopy.
- Dedicated BL2+ tissue culture suite with two large incubators, two 4-ft hoods, two large centrifuges in the room, a fluorescence digital inverted microscope with four color channels (EVOS), an electroporator (Neon), and an automated fluorescence cell counter (Countess). Access to various fluorescence, multiphoton, and confocal microscopes, flow cytometry, cell sorting, and histology core facilities within the building.
- Genomics: Genome editing with CRISPR/Cas9, mutagenesis and mutation detection, DNA isolation, Sequencing, DNA library preparation for NGS.
- A shared pyrosequencer. Immediate access to various RNASeq and NGS equipment. Bioinformatics and DNA sequencing core facilities within the building.
- Animals: Currently animal research has not yet started in the lab. However, Dr. Jain has experience in mice breeding, genotyping, tagging, injections, surgeries, perfusion, organ isolation, magnetic cell isolation, IVIS imaging, and euthanasia.
- Jain lab owns a Miltenyi’s autoMACS Pro Separator, an isoflurane device, a tissue homogenizer (coming soon). The Jain lab is located in the Cancer and Genetics Research Complex, which has an animal facility within the building.
- Computer and software: Dedicated lab server and a computer for individuals and equipment in the lab (>10 total). Softwares for bioinformatics, plasmid design, molecular modeling, graphical design, experimental design, and statistical analysis are available in the lab.