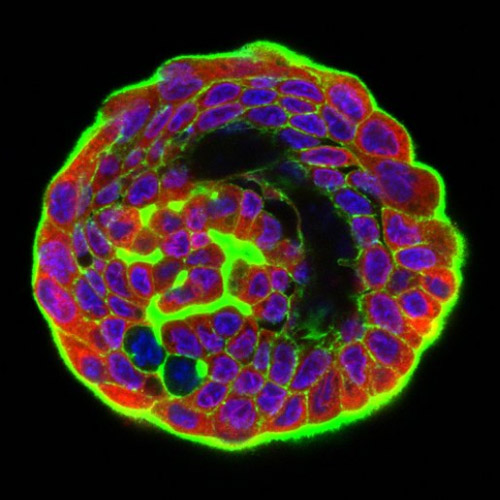
Mechanobiology
We study coupled mechanical and biochemical changes in human disease

Principal Investigator
Dr. Tanmay Lele, Ph.D.
Professor
What is mechanobiology?
Our research is in the area of Mechanobiology. Mechanobiology is the scientific study of the mechanisms by which cell generated mechanical forces and/or the cellular mechanical environment impact cell and tissue structure and function. We are particularly interested in the mechanobiology of human diseases including cancer.